|
This superfamily, as currently recognised, groups together many
families of large moths mainly on characters of absence (Common,
1970; Franclemont, 1973) : of the M vein stem in the cells of
both wings (though this is present in the forewings of some
genera in the Bombyx lineage of the Bombycidae, and the
hind-wing of some Brahmaeidae); of the tongue and frenulum
(though the former is strongly developed in many Sphingidae, as
is the latter (also in Apatelodidae), and the frenulum is weakly
present in other families, e.g. Bombycidae).
The strongly bipectinate male antennae in most families, associated with
strong sembling behaviour to female pheromones and the spinning
of a fine silken cocoon, though widely distributed and highly
developed in the Bombycoidea, are not unique to them: the
Lymantriidae, in the Noctuoidea, also have strongly bipectinate
antennae in the male, sembling behaviour to a static female, and
lack a tongue. Lack of adult feeding and male sembling to a
static female may be biologically linked, as described for the
Saturniidae by Janzen (1984).
On these characters alone, therefore, there is no indication the
superfamily is monophyletic: a natural grouping.
However, there are a number of features that link the families, or a
larger portion of them, together and indicate a relationship
with the Neotropical Mimallonoidea (Mimallonidae), treated as a
separate superfamily by Franclemont (1973). In the Mimallonoidea
ocelli, tongue and palps are absent, and the larva is striking
in that later instars build a portable case of silk
incorporating fragments of vegetation and frass.
The families of Bombycoidea represented in South-East Asia are the
Lasiocampidae, Eupterotidae, Bombycidae, Brahmaeidae,
Saturniidae and Sphingidae plus a new, anomalous taxon, Kenguichardia gen.
nov., tentatively associated with the Bombycoidea by the loss of
the stem of vein M in the wing cells, lack of tympanal organs,
and presence of the character of the forewing radial veins
discussed in the next section. Tongue and frenulum are strong,
though ocelli and chaetosemata are absent.
Other bombycoid families are the Australasian Anthelidae and
Carthaeidae, the Palaearctic Lemoniidae, Mirinidae (Minet, 1986)
and Endromidae, and the New World Oxytenidae and Cercophanidae
(allied to Saturniidae), and Apatelodidae. The South African
genus Spiramiopsis Hampson is of uncertain placement,
though currently resting in the Bombycidae (Fletcher & Nye,
1982), and has a number of interesting features.
WING
VENATION
The absence, and occasional presence, of the M stem in wing cells has
already been mentioned. One feature common to many bombycoid
lineages is the independent stalking of the second and third
radial veins of the forewing. This is seen in some homoneurous
non-ditrysian groups such as the Hepialidae, but is very rarely
found in Ditrysia (present in Dalceridae).
To illustrate the distribution of this feature simply amongst the
various bombycoid groups, the configuration of the radial veins
as they arise from the cell can be expressed by means of a
shorthand system. A semicolon separates veins or branching
systems that arise independently from the cell; where they are
connate (not stalked but arising from the same point on the
cell) a hyphen is used. A system of brackets can be used to
indicate order of separation of veins in a branching system and
whether they are anterior or posterior. For example, the
Megalopygidae are distinguished from the Limacodidae in the key
to families in part 1 of this series (Holloway,1986) by whether
R4 arises anteriorly or posteriorly from Rs. This can be
expressed as (R2(R3(R4,R5))) for Megalopygidae and
(R2((R3,R4)R5)) for Limacodidae, those veins listed after the
comma arising posteriorly, those before anteriorly, and the
order of bracketing from the distal dichotomy indicating the
order of branching off from Rs basad. A fusion, e.g. in the
hindwing of Brahmaeidae, can be indicated as R1>Sc, and an
anastomois (fusion then separation), as in the formation of the
forewing areole in the Anthelidae, as R3><R4, or, as in
formation of the Dalceridae areole (Holloway, 1986,fig.13), R2 +
3><R4 + 5.
When the number of radial veins is reduced, it is sometimes apparent
which have become fused or lost, as in the Sphingidae, but often
this not the case. In the latter instance it is perhaps least
confusing to leave the R veins unnumbered.
The
following examples illustrate some of the variety of venation
found in the Bombycoidea, from groups with a full complement of
veins to those with one or two of the radials lost. Note the
recurrence of R2, R3 in the groups with all
radials present except for the Bombyx lineage of the
Bombycidae and the Apatelodidae:
|
(R1); (R2, R1); ((R4, R5)M1)
or (R4, R5)-(M1)
|
Mimallonidae
Lasiocampidae: Alompra group
Sphingidae: some Smerinthini
|
|
(R1); (R2 + 3); R4, R5) - (M1)
|
Most Sphingidae
|
|
(R1); (R2, R3); (R4(R5, M1))
|
Most Lasiocampidae
|
|
(R1); ((R2, R3)R4); (R5, M1)
|
Lasiocampidae: Bhima
|
|
(R1); (R2, R3); (R4, R5); (M1)
|
Carthaeidae
|
|
(R1); (R2, R3); (R4, R5) - (M1)
|
|
|
R3><R4
|
Anthelidae
|
|
(R1); (R2, R3); (R4); (R5); (M1)
|
Kenguichardia
|
|
(R1)-(((R2, R3)R4)R5)) - (M1)
|
Acanthobrahmaea
|
|
(R1); ((R2, R3)(R4, R5)) - (M1)
|
Brahmaea
|
|
(R1); ((R2(R3(R4, R5)))M1)
|
Bombycidae: Bombyx group
|
|
(R1); ((R2(R3(R4, R5))) - (M1)
|
Apatelodidae: Apatelodes
|
|
(R1); ((R, R)R) - (M1)
|
Spiramiopsis
|
|
or (((R, R)R)M1)
|
Lemoniidae: Lemonia
Bombycidae: Mustilia group
Eupterotidae
Saturniidae: Citheronia
|
|
((R1(R, R))R); (M1)
|
Saturniidae: Actias, Loepa, Cricula
|
|
(R1); ((R, R) R); (M1)
|
Saturniidae: Samia, Attacus
Endromidae
|
|
(R1(R, R)); (M1)
|
Saturniidae: Antheraea
|
|
(R1) - ((R,R)M1)
|
Saturniidae: Anisota
|
|
(R1); (R, R); (M1)
|
Saturniidae: Hemileuca
|
RESTING
POSTURE OF THE ADULT
Broad, deep-winged groups such as most Saturniidae,
the Eupterotidae and
Brahmaeidae rest with
the wings flat and both wings exposed. Therefore the hindwing is
usually as strongly and extensively patterned as the forewing. Lasiocampidae
rest with the wings steeply tented over the body, the expanded
humeral area of the hindwing protruding from under the costa of
the forewing, giving a very disrupted, irregular outline that
combines with cryptic patterning to give a dead leaf effect. In
most Lasiocampidae the hindwings are most strongly patterned
costally because of this posture. Sphingidae:
Macroglossinae have a swept back, jet fighter type of posture.
The tribe Sphingini of the Sphinginae hold their wings at a more
swept back angle, and angled towards the substrate, usually a
tree trunk; the forewing pattern is often patterned for crypsis
on bark, sometimes with flash coloration on the hindwing. None
of these indicate any linkage between bombycoid families.
However,
some of the Sphinginae: Smerinthini (eg. Smerinthus,
Daphnusa), the Bombycidae,
the South Africa Spiramiopsis (N. Duke, pers. comm.)
and Apatelodidae share a peculiar resting posture not
seen in other macrolepidoptera. The forewings are held out at
right angles to the body, with strongly patterned dorsum of the
hindwing scrolled up and over that of the forewing. The
abdomen is usually flexed upwards or sideways.
MALE
GENITALIA
In
most groups the uncus of many of the taxa is bifid or bilobed;
when entire, as in most Sphingidae: Macroglossinae, this appears
to be a derived character.
Kuznetzov
& Stekolnikov (I985) studied genital musculature in several
of the bombycoid families, concluding they were
interrelated and suggesting the interrelationships were as in
Fig. 1. All except the Lasiocampidae share a secondary type of
muscle insertion. These authors split the superfamily into the
three: Lasiocampoidea, Sphingoidea and Bombycoidea sensu
stricto.
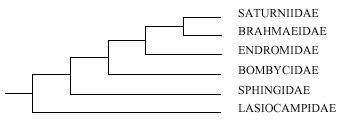
Figure 1. Diagram
of relationships of some Bombycoidea families according to
Kuznetzov & Stekolnikov (1985).
LARVAE
The generalised condition of a macrolepidopteran
larva might be taken to be possession of three pairs of verrucae
or scoli on each segment, invested with secondary setae. These
are situated dorsally, subdorsally, and laterally below the
spiracles. This condition is seen in many Saturniidae and in the
verrucae, when present, of Lasiocampidae.
In all Bombycoidea there are secondary setae not
associated with verrucae or scoli, often very numerous, and with
the superfamily there is a trend either towards great hairiness
(Lasiocampidae, Eupterotidae, Anthelidae, Lemoniidae some
Apatelodidae, some Saturniidae), or reduction of the setae to
fine spines or granulation, and associated reduction of the
scoli in number, yet often with enlargement of a
few of them (other families, a few Apatelodidae and many
Saturniidae). In all these families with non-hairy larvae, the
eighth abdominal segment has a central scolus or horn
dorsally rather than a pair, though some Saturniidae (especially
Saturniidae) have a pair or show signs of partial fusion of a
pair (W. Nassig, pers. comm.). Most Bombycidae,
the Sphingdae, a few Apatelodidae,
the Oxytenidae, Endromidae and early instars of the Carthaeidae
have the caudal horn only. In Bombycidae the thoracidae segments
are not ornamented; dorsal pairs of filaments occur on segments
A1-7, A9 in one species of Bombyx and members of the Ocinara
group and Gunda often have transverse swellings
dorsally on A2 and A5. However, many other groups have paired
filaments or horns on T2 and T3 as well as the horn on A8 but rarely
on other abdominal segments, such as the Brahmaeidae (a small
pair also on A9), Spiramiopsis, several subfamilies of
the Saturniidae (Agliinae, Rhescyntinae, Citheroniinae); the
Cercophanidae have caudal and thoracic areas produced into
tapering processes. Thus there are rather unusual larval
features linking many of the bombycoid families. Snake mimicry
is also a recurrent feature (Macroglossinae, final instar
Brahmaeidae, some Bombycidae).
Larvae
of Sphingidae, Saturniidae and Brahmaeidae have a 'sphinx-like'
alarm posture with head and thoracic segments retracted and
lifted off the substrate; the dorsum of the thorax appears
expanded and directed forwards.
Thus,
with unusual features of venation, resting posture, male
genitalia and larvae found to be common to diverse families
within the Bombycoidea, there are some grounds for considering
that the group may be a natural one (see also Minet (1986)). The
diagram of family relationships in Fig. 1 would require reversal
of the dorsal pair of scoli on A8 in some lineages of
Saturniidae.
HOST-PLANT
RELATIONSHIPS
Host-plant
specialisation at the family level is only seen in some of the
smaller Bombycoid families, with the Brahmaeidae mostly recorded
from Oleaceae, the Bombyx lineage of the Bombycidae from
Moraceae and the Mustilia lineage from the Symplocaceae
and the Theaceae, two closely related families. In the
Eupterotidae the genus Ganisa has only been recorded from
Oleaceae, as has the genus Dolbina in the Sphingidae.
Also amongst the Sphingidae, the genera Clanis and Sataspes
would appear to be restricted to Leguminosae.
Most
taxa in the other families appear to have a range of diet
covering several plant families. It is instructive to note the
frequency with which various families have been recorded as
host-plants in the various major bombycoid groups. Janzen (1984)
drew attention to dietary differences between the Saturniidae
and Sphingidae in seasonal forest in Costa Rica. But there are
even more striking differences between the major groupings with
the Sphingidae. There is no dietary overlap between the
Sphingini (12 spp), Smerinthini (3 spp) and
Macroglossinae (17 spp), as can be seen from the Table 1 below,
illustrating just the more frequently recorded families.
There
is a very similar dietary segregation within the Bornean
sphingids (Table 2), each subfamily having much the same plant
family preferences as in Costa Rica, and only slight overlap
between the groups, eg. Leguminosae in Sphingini and
Smerinthini and Convolvulaceae for Sphingini and Macroglossinae.
The Smerinthini are much more diverse in Borneo than Costa Rica.
Table 1.
Most frequently recorded host-plant families for Sphingidae
higher taxa in Costa Rica; data from Janzen (1984). The number
of genera in each family recorded for each sphingid species are
summed for all species (i.e. the same plant genus will be
counted more than once if fed on by more than one
species).
|
Sphingini
|
Smerinthini
|
Macroglossinae
|
|
Bignoniaceae
(10)
|
Anacardiaceae
(2)
|
Rubiaceae
(22)
|
|
Verbenaceae
(5)
|
Lauraceae
(2)
|
Dilleniaceae
(5)
|
|
Compositae
(5)
|
|
Apocynaceae
(5)
|
|
Solanaceae
(5)
|
|
Moraceae
(4)
|
|
Convolvulaceae
(4)
|
|
Vitidaceae
(3)
|
|
Annonaceae
(2)
|
|
Euphorbiaceae
(2)
|
|
Boraginaceae
(2)
|
|
Asclepiadaceae
(1)
|
|
Labiatae
(2)
|
|
Sapotaceae
(1)
|
Table 2.
Most frequently recorded host-plant families for Bornean
Sphingdae, collated from the full geographical range of the
species and presented as in Table 1.
|
Sphingini
|
Smerinthini
|
Macroglossinae
|
|
Bignoniaceae
(18)
|
Leguminosae
(13)
|
Araceae
(40)
|
|
Oleaceae
(17)
|
Fagaceae
(8)
|
Vitidaceae
(38)
|
|
Verbenaceae
(16)
|
Anacardiaceae
(6)
|
Rubiaceae
(29)
|
|
Leguminosae
(15)
|
Sterculiaceae
(4)
|
Dilleniaceae
(10)
|
|
Labiatae
(5)
|
Bombacaceae
(4)
|
Onagraceae
(8)
|
|
Convolvulaceae
(5)
|
Lauraceae
(3)
|
Leeaceae
(7)
|
|
Scrophulariaceae
(4)
|
Burseraceae
(2)
|
Convolvulaceae
(6)
|
|
Pedaliaceae
(3)
|
Sapindaceae
(2)
|
Balsaminaceae
(6)
|
|
Malvaceae
(2)
|
Guttiferae
(2)
|
Actinidiaceae
(6)
|
|
Meliaceae
(2)
|
|
Nyctaginaceae
(5)
|
|
Araceae
(2)
|
|
Polygonaceae
(4)
|
|
Caprifoliaceae
(2)
|
|
Begoniaceae
(3)
|
There are some major differences however, that may reflect
ecological differences between the two regions. Also the host
data for the Bornean species are collated from the whole
Indo-Australian ranges of many of the species, but those for
Costa Rica are from a very restricted locality. Oleaceae are
strongly represented for the Sphingini, Leguminosae for
Sphingini and Smerinthini, Fagaceae for the latter, and Araceae
and Vitidaceae for the Macroglossinae. These plant families are
utilised only infrequently or not at all in Costa Rica.
The
other major group considered by Janzen was the Saturniidae
contrasting both their host-plant range and adult behaviour with
that of the Sphingidae. Many saturniid subfamilies are
represented in Costa Rica, but only the Saturniinae in Borneo.
The host-plant data for this family are presented in Table 3.
Table 3.
Most frequently recorded host-plant families for Saturniidae in
Costa Rica, and for Saturniinae (CR) and Borneo (B), compiled as
described for Tables 1 and 2.
|
Saturniidae
(CR)
|
Saturniinae
(CR)
|
Saturniinae
(B)
|
|
Leguminosae
(34)
|
Rubiaceae
(3)
|
Rosaceae
(14)
|
|
Rubiaceae
(9)
|
Anacardiaceae
(3)
|
Lauraceae
(8)
|
|
Flacourtiaceae
(7)
|
Flacourtiaceae
(2)
|
Anacardiaceae
(7)
|
|
Bombacaceae
(5)
|
Rutaceae
(1)
|
Rutaceae
(6)
|
|
Sapindaceae
(5)
|
Lauraceae
(1)
|
Euphorbiaceae
(5)
|
|
Sterculiaceae
(5)
|
|
Meliaceae
(5)
|
|
Anacardiaceae
(4)
|
|
Fagaceae
(4)
|
|
Fagaceae
(3)
|
|
Lythraceae
(4)
|
|
Myrtaceae
(3)
|
|
Simaroubaceae
(4)
|
|
Rutaceae
(3)
|
|
Rhamnaceae
(4)
|
|
Bignoniaceae
(3)
|
|
Leguminosae
(3)
|
|
|
|
Corylaceae
(3)
|
The
six most popular families are not the same in the two regions,
but overall four families are shared, and three families between
the Saturniinae in each locality.
Janzen
(1984) divided the Costa Rica 'big moths' of the Bombycoidea
into two behavioural categories: (1) those that do not
feed as adults are shortlived, the male searching for a static
female calling with a pheromone, have marked sexual dimorphism,
oviposit in masses, usually in tree crowns, the larvae
aposematic or mimicking urticating forms, feeding on foliage
often low in nutrients, high in phenolics (Janzen &
Waterman, 1984); (2) both sexes feeding, highly active,
streamlined, long-lived with sexual dimorphism weak or absent,
ovipositing singly on small plants, saplings, shrubs, vines or
tree crowns, the larvae cryptic, with a high rate of nutrient
assimilation from young or old foliage that is more nutrient
rich though often containing toxic alkaloids, but being in low
in phenolics (Janzen & Waterman, 1984).
Though
the Sphingini and Macroglossinae of Costa Rica and Borneo show
close parallels, the representation of weak-tongued Smerinthini
and other non-feeding big moths is somewhat different, with a
very diverse saturniid fauna in Costa Rica, but only weak
representation of Smerinthini and, for the Neotropics as a
whole, Lasiocampidae. In Borneo the Smerinthini and
Lasiocampidae are diverse, Eupterotidae are present, yet the
Saturniidae are only weakly represented and that by only one
subfamily. It could therefore be that the non-feeding 'big moth'
niche is occupied by Lasiocampidae, Eupterotidae, Saturniinae and
Smerinthini for Borneo. This contention is supported when the
host-plant preferences of Eupterotidae and Lasiocampidae are
added to those of Sarturniinae and Smerinthini for Borneo and
compared with those for Saturniidae plus Smerinthini for Costa
Rica. (Table 4). There are nine families in common, with
Rubiaceae, Flacourtiaceae and Bignoniaceae peculiar to Costa
Rica, and Rosaceae, Meliaceae, Combretaceae, Lythraceae and
Rhamnaceae to Borneo.
Table 4. Most
frequently recorded host-plants for non-feeding 'big moths' in
Costa Rica (NFBM - CR) and Borneo (NFBM - B), calculated as
discussed in Tables 1 and 2.
|
Lasiocampidae
|
NFBM
- CR
|
NFBM
- B
|
|
Leguminosae
(15)
|
Leguminosae
(34)
|
Leguminosae
(35)
|
|
Myrtaceae
(15)
|
Rubiaceae
(9)
|
Rosaceae
(18)
|
|
Combretaceae
(5)
|
Flacourtiaceae
(7)
|
Fagaceae
(16)
|
|
Euphorbiaceae
(4)
|
Anacardiaceae
(6)
|
Lauraceae
(16)
|
|
Lauraceae
(3)
|
Sapindaceae
(5)
|
Anacardiaceae
(14)
|
|
Sapotaceae
(3)
|
Sterculiaceae
(5)
|
Rutaceae
(9)
|
|
Theaceae
(3)
|
Lauraceae
(4)
|
Meliaceae
(8)
|
|
Verbenaceae
(3)
|
Fagaceae
(3)
|
Combretaceae
(8)
|
|
Rosaceae
(3)
|
Myrtaceae
(3)
|
Lythraceae
(7)
|
|
Rutaceae
(3)
|
Rutaceae
(3)
|
Bombacaceae
(6)
|
|
Dipterocarpaceae
(3)
|
Bignoniaceae
(3)
|
Sterculiaceae
(6)
|
|
|
|
Rhamnaceae
(6)
|
|
|
|
Sapindaceae
(5)
|
|
Eupterotidae
|
|
|
|
Leguminosae
(4)
|
|
|
|
Lauraceae
(2)
|
|
|
|
Oleaceae
(2)
|
|
|
|
Combretacea
(2)
|
|
|
Whilst
all Bornean Saturniinae are deep-winged, Costa Rica Saturniidae
have a diversity of wing forms. However, the inclusion of
Eupterotidae and Lasiocampidae as Bornean non-feeding big moths
brings the Bornean situation into closer parallel with that in
Costa Rica. The similarity of the Lebeda to Kunugia
group of lasiocampid genera in form to that of the Costa Rican
Ceratocampinae is particularly striking.
<<Return to Contents page
|